Delivering the Right Antibiotic,
at the Right Time, Every Time.
Same-shift AST results within 4 hours.
 For Research Use Only (RUO)
For Research Use Only (RUO)Same-day results in just 72 hours
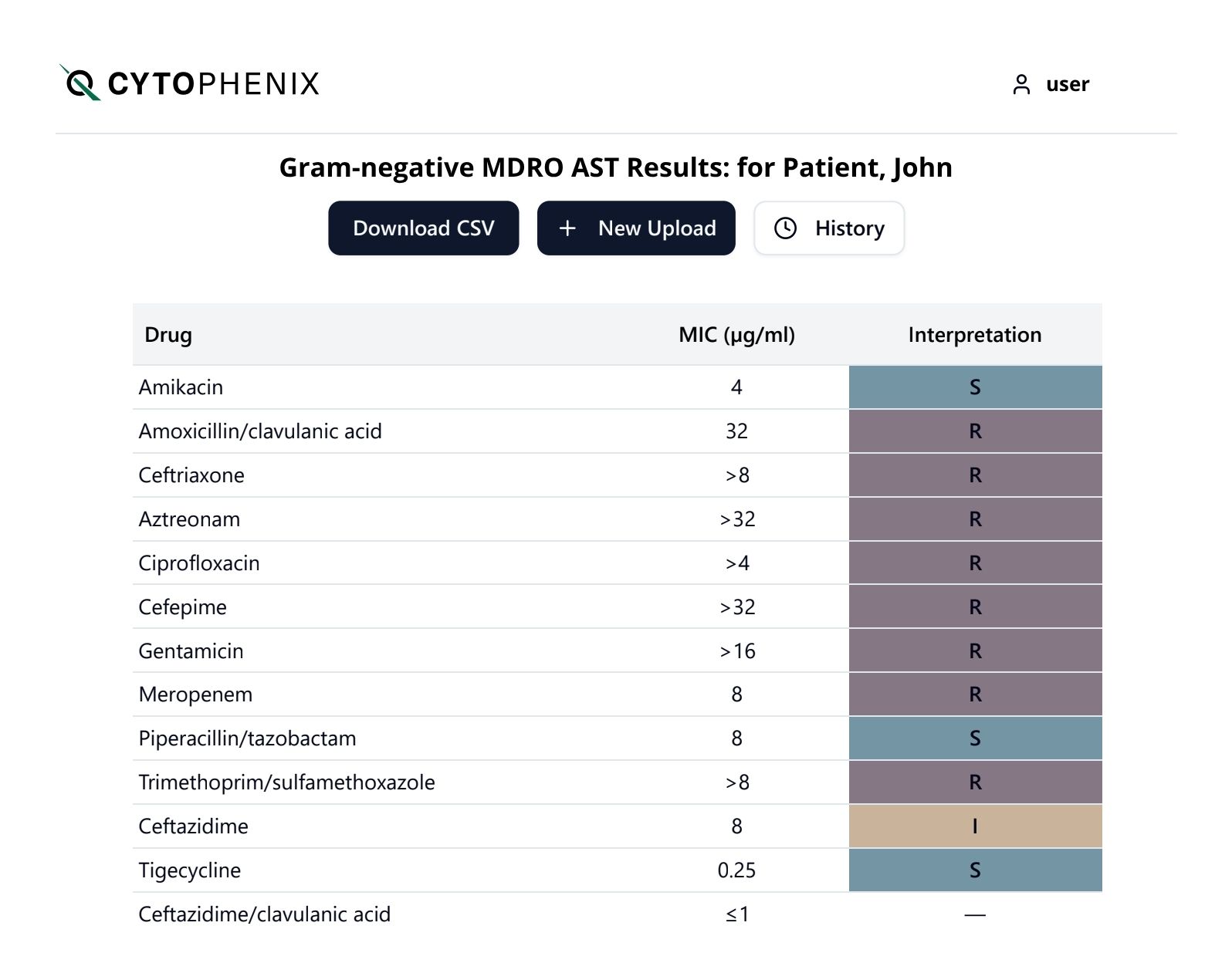
Cytophenix FloCAST™ provides same-day results in just 4 hours, delivering clinical confidence for doctors making antibiotic treatment decisions from 48-72 hours faster than available methods.
Cytophenix FloCAST™ combines the promise of AI enabled interpretation with acoustic flow cytometry to provide single cell resolution of the real-time response of bacterial and fungal cells to a complete panel of antimicrobial agents, including antibiotics and antifungals.
Learn More Critical
Unmet Needs
in Sepsis Treatment
Doctors rely on the Antimicrobial Susceptibility Test (AST) for making definitive treatment decisions for patients with infectious diseases. Without prompt and effective treatment, infections can progress to life-threatening sepsis. Yet AST methods have remained largely unchanged for nearly a century.
Laboratory automation solutions reduce labor inputs — without meaningful improvement in clinical outcomes. Doctors wait from 3–5 days from onset of symptoms to receive critical diagnostic data — the AST result — to guide the definitive treatment decision.
Doctors manage this clinical ambiguity with empiric antimicrobial therapy — a 'best guess' — with multiple powerful antimicrobial treatments concurrently administered. Frequently, this includes two or more antibiotics, and may include concurrent antifungals, as doctors balance immediate risk from sepsis with delayed risks from extended broad-spectrum antimicrobial treatment. Mortality risks increase by up to 7.6% for each hour effective treatment is delayed. (Kumar et al.)1 (Liu et al.)2
Sepsis is most commonly the result of untreated or undertreated serious infections. It continues to kill 11 million individuals globally with a staggering cost of care of $2.4 trillion (USD) annually.
receive ineffective empiric antimicrobial therapy
(Kadri et al.)3
receive effective but excessive antimicrobial therapy
(Liu et al.)4
Transforming
Infection Diagnosis
Cytophenix is reshaping the established paradigm of the care and management of infectious diseases leading to sepsis.
Our AI-powered platform Cytophenix FloCAST™ accelerates AST results from the laboratory and delivers clinical confidence to doctors 48-72 hours faster than existing methods.
Equipped with AST results in just 4 hours, doctors can review antimicrobial treatment decisions with clinical certainty: escalate, de-escalate, or confirm antibiotic therapy.
Cytophenix FloCAST™ is for microbiologists, pharmacists, antimicrobial stewardship teams, and doctors serving critically-ill patients at risk of sepsis.
Learn About Sepsis
Advancing Innovation in Antimicrobial Stewardship
Antimicrobial stewardship teams are under myriad pressures. At the patient bedside, they meet the urgent need to treat serious infections effectively.
At the same time, members of antimicrobial stewardship teams face significant administrative demands. Executive leaders hold them accountable for goals tied to the reduction of unnecessary antimicrobial use, improving clinician education, and documentation of impact.
Cytophenix FloCAST™ can help. The Cytophenix FloCAST™ platform is for microbiologists, pharmacists, and doctors serving critically-ill patients at risk of developing sepsis. And it is more than that.
Accelerating antimicrobial therapy decisions with fast AST results reduces the overall antimicrobial load on patients, thus reducing the evolutionary pressures that lead to antimicrobial resistance. Same-shift AST results thus spare patients lives, and preserve antibiotics and antifungals for our children and grandchildren.

Capital-Efficient.
Accelerated Development.
Life-Saving Diagnostics.
We reduce R&D footprint through a development partnership with an established flow cytometry global manufacturing leader. The validated technology platform supports open architecture for novel applications, and the development partner's commercial relationships for distribution are deeply embedded into US healthcare.
As a result, investment dollars are efficiently targeted for groundbreaking assay, data science, and software development, rather than large-scale manufacturing capital expenditure.
Founder-Led.
Research-Driven.
Cytophenix is a founder-led biotechnology company advancing rapid diagnostics for bloodstream infections. We are based in Western Australia's research corridor in Perth. Our team members include many with dual appointment at world-renowned research facilities The Harry Perkins Institute and The University of Western Australia.
With over 85% founder ownership and $1.3 million pre-seed raised, our team maximizes current and subsequent investor capital.

As of March 2026, our first US Key Opinion Leader pilot site is online in a Top 100 academic health care facility on the West Coast.
Meet the TeamReferences
- Kumar et al. 2006, "Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock" PMID: 16625125.
- Liu et al. 2017, "The Timing of Early Antibiotics and Hospital Mortality in Sepsis". PMID 28345952.
- Kadri et al. 2021, "Inappropriate empirical antibiotic therapy for bloodstream infections based on discordant in-vitro susceptibilities: a retrospective cohort analysis of prevalence, predictors, and mortality risk in US hospitals".
- Liu et al. 2016, "Frequency of empiric antibiotic de-escalation in an acute care hospital with an established Antimicrobial Stewardship Program".